Philips BiPAP Cancer
FDA Philips Warning for CPAP Cancer
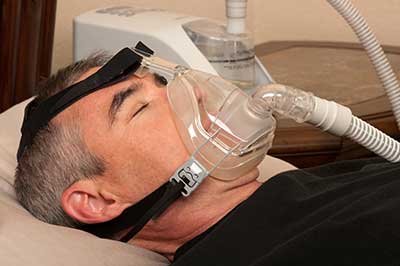
Philips BiPAP machines have been found to cause cancer and have been recalled across the globe as a result. A foam component found in Philips CPAP and BiPAP machines as well as life-supporting mechanical ventilators is prone to degradation over time and in certain environmental conditions. When the foam breaks down, toxic particles and gases are released into the device's airstream and enter the patient's airway. Patients who can stop using their machines immediately are advised to do so to avoid further exposure to the risk of Philips CPAP cancer. Lawyers handling Philips CPAP cancer lawsuits believe persons and family members of persons who have developed cancer or another serious side effect after using a machine from the Philips BiPAP recall may be eligible for significant compensation. This page provides a comprehensive look at the Philips CPAP cancer risks and warnings.
FDA Warns Philips CPAPs, BiPAPs and Mechanical Ventilators Cause Cancer
Royal Philips, a multinational medical technology corporation based in the Netherlands manufactures a broad range of medical devices. This product line includes several different types of breathing machines used to treat sleep apnea and provide mechanical life support. Specifically, the devices included in the Philips BiPAP / CPAP cancer recall include CPAP (continuous positive airway pressure) machines and BiPAP (bilateral positive airway pressure) machines for sleep apnea, as well as life-supporting mechanical ventilators.
When a foam part in Philips BiPAP machines breaks down, toxic particles and gases are released into the device's airstream and enter the patient's airway. Patients who can stop using their machines immediately are advised to do so to avoid further exposure, according to the FDA Philips CPAP Cancer Warning.
The public's first inkling of the Philips BiPAP cancer risk was in April of 2021, when Philips first alluded to a "quality issue" that had been detected with some of its CPAP, BiPAP and mechanical ventilator machines in a shareholder communication. Prior to that announcement, Philips executives had received reports from the field citing a risk of Philips BiPAP cancer from the machines. Patients were reportedly becoming ill when a foam component in the machine (used to minimize the machine's noise) began to break down.
FDA Philips BiPAP Cancer Warning
More than a month later, Philips finally issued a global recall, citing the risk of cancer from Philips CPAP use. The Philips BiPAP cancer recall concerned 3-4 million machines in use worldwide, all of which included the foam component in question.
The part, which was referred to as "sound abatement foam", is made of PE-PUR, or polyester-based polyurethane. With age and exposure to environmental heat and dampness, the PE-PUR foam part can start to degrade, releasing toxic gases and small particles into the CPAP machine's airflow. Persons using the machines inhale these dangerous particles and are put at risk for serious side effects including BiPAP cancer.
According to the Philips CPAP cancer recall, exposure to chemical emissions has been found to cause the following adverse effects:
- Risk of cancer, or "toxic and carcinogenic effects"
- Headache and dizziness
- Irritation of the eyes, skin and respiratory tract
- Hypersensitivity
- Nausea and vomiting
The factors that affect the breakdown of this foam substance to the point that Philips BiPAP machines cause cancer include the passing of time, environmental heat and humidity, and the use of unapproved cleaning methods such as ozone.
Who can file a ventilator recall lawsuit?
Patients who have used one of more than twenty different types of breathing machines included in the Philips CPAP, BiPAP and ventilator recall and then developed cancer or another serious medical problem may be eligible to file a claim against the Dutch manufacturer. Click here to learn more.
The following devices are included in the Philips BiPAP cancer recall:
- All Philips CPAP devices manufactured before April 26, 2021, under all serial numbers
- All Philips BiLevel PAP devices manufactured before April 26, 2021, under all serial numbers
- E30 model continuous ventilator, minimum ventilatory support for facility use
- DreamStation non-life supporting continuous ventilator models ASV, ST, and AVAPS
- SystemOne ASV4 model continuous ventilators, non-life supporting
- C Series continuous ventilator, non-life supporting models ASV, S/T, and AVAPS
- OmniLab Advanced Plus in-lab titration device
- SystemOne Q series models of non-continuous ventilators
- DreamStation CPAP, Auto CPAP, and BiPAP ventilator models
- DreamStation Go CPAP and APAP models of non-continuous ventilators
- Dorma 400 and 500 CPAP models of non-continuous ventilators
- REMStar SE Auto CPAP models of non-continuous ventilators
- The Trilogy 100, Trilogy 200, Garbin Plus, Aeris, and LifeVent continuous ventilators
- The A-Series BiPAP V30 Auto and Hybrid A30 continuous ventilators for minimum ventilatory support for facility use
- Outside the United States, the A-Series BiPAP A30 and A40 non-life supporting continuous ventilators
The FDA Philips BiPAP cancer warning indicates that wherever possible, patients utilizing the recalled devices should be moved to a different machine.
Philips CPAP Sleep Apnea Machines Cause Cancer
The majority of the devices included in the BiPAP cancer recall are CPAP or bilateral PAP machines used to treat sleep apnea. Sleep apnea is a breathing disorder that puts approximately 20 million Americans at risk for stroke, heart problems and premature death. Sleep apnea machines dramatically improve sleep and reduce possible health risks. The problem with Philips sleep apnea machines is that many have been found to cause cancer. In other words, millions of patients who are using these devices ro reduce their exposure to one set of serious health problems, are now learning they have potentially been exposed to toxic and cancer-causing chemicals each and every night.

Countless patients have relied on Philips CPAP machines for sleep apnea over the course of many years or even decades with no warning of the BiPAP cancer risk from Philips sleep apnea machines. The Philips CPAP cancer warning states that anyone who is still using a recalled Philips CPAP machine or BiPAP machine halt use of the device immediately due to the Philips CPAP cancer risk.
Philips Mechanical Ventilators Cause Cancer
Of the 3-4 million devices included in the Philips cancer recall, 20% are life-supporting mechanical ventilators. While especially vital in the ongoing global fight against COVID-19, mechanical life supporting ventilators have long been an essential medical device that enables life-saving surgeries and healing time for the most severely ill or injured patients. It is horrifying to learn this vital piece of equipment used with the most vulnerable patients actually emits toxic and cancer-causing gases and particles.
Let Our BiPAP Cancer Attorneys Help You
Our lawyers specialize in holding large corporations accountable when they've placed profits ahead of safety. Through settlements and winning verdicts, our attorneys have obtained millions for our clients. Let us help you today.
Philips CPAP Cancer Lawsuits
Filing a lawsuit will allow you to hold the pharmaceutical company accountable for damage it has caused you or a loved one, while also providing real compensation for your medical expenses, suffering and loss. Contact us today for a free consultation.








